From Concept to Product to Market
From Concept…
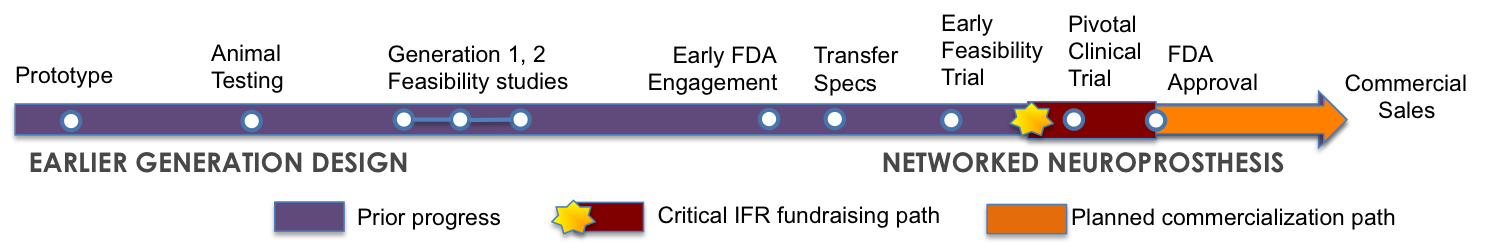
The Networked Neuroprosthesis is a third-generation design that started out in 1999 as sketches on a white board. Based on their past experience, our engineers imagined new ways to design a neuromodulation system that would permit multiple functions from a small set of building blocks.
We threw out early concepts that would have required attendants to place external components, in favor of a completely implantable system that a person can use independently.
…To Product…
Between 2002 and 2011, the IFR team developed each of the basic components of the system and conducted engineering analyses. To date we have filed and received numerous patents for the Networked Neuroprosthesis. By 2007, we began turning our early prototypes into devices, an effort that continues today with our manufacturing partner, Synapse Biomedical. The first working prototype for the complete system was developed and tested in 2012.
All of these efforts have been supported by funding from the NIH (National Institute for Neurological Diseases and Stroke, and National Institute for Biomedical Imaging and Bioengineering), the Veterans Administration, and the Paralyzed Veterans of America, totaling $15 million.
In 2014 and 2015, with philanthropic support, we applied for and received full approval from the FDA to begin evaluating the Networked Neuroprosthesis to restore both hand function and trunk stability in an early feasibility trial ten participants with tetraplegia.
Our next goal is to raise the funds necessary to conduct a pivotal clinical trial in multiple centers in order to collect additional evidence for safety and effectiveness. Then we will file for FDA market approval.
…To Market!
The Networked Neuroprosthesis is designed to restore multiple functions, each of which is being developed by a different team of investigators. Many of these teams have taken their earlier generation designs well into development and have demonstrated functional restoration in dozens of participants.
These teams cannot bring these early systems through the full commercialization process alone, and this is where IFR steps in. We serve as a partner, raising funds for clinical trials, navigating the regulatory requirements, and shepherding the systems through to market with the help of our industry partner.
Funding for the IFR helps support these final commercialization steps.